- Serology
Western blotting:
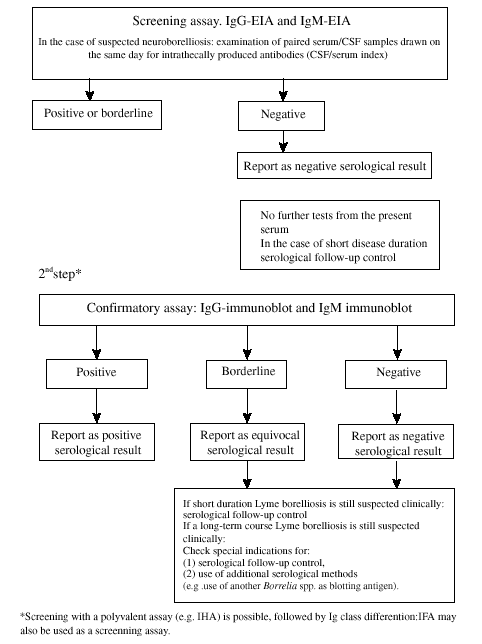
Fig. 7. Stepwise serodiagnosis of Lyme borelliosis [92]. IFA, immunofluorescent assay; IHA, immunohaemagglutination assay.
[Comment by J. Gruber:
As in most other similar reviews, a rational is not given here for requiring consecutive tests, thereby reducing the chance of detecting the illness. The recommendation of this "two-tier-testing" sheds a strange light on the reliability of the first test in the series.
See also: Marangoni A et al., Comparative evaluation of three different ELISA methods for the diagnosis of early culture-confirmed Lyme disease in Italy, J Med Microbiol 2005 Apr 54(Pt 4):361-7:The sensitivities of the ELISAs were as follows:
- Enzygnost Borreliosis IgM , 70.5 %;
- Quick ELISA C6 Borrelia, 62.1 %;
- recomWell Borrelia IgM, 55.7 %;
- recomWell Borrelia IgG, 57.9 %; and
- Enzygnost Borreliosis IgG, 36.8 %]
In order to interpret the individual reactivity patterns of patient sera correctly, it is necessary to be familiar with the stage dependent diagnostic significance of immunogenic proteins. For example, in the diagnosis of early Lyme borreliosis, the intensity of at least some of the immunoblot bands (p41, OspC) must be taken into account.
Therefore, each assay requires precise internal controls that allow semiquantitative evaluation of blot intensities.
- Wholecell antigen assays are available, in which electrophoretically separated wholecell lysates from borreliae are employed, with the borrelial proteins transferred to blot membranes.
- Suppliers also offer recombinant antigen assays in which, analogously, selected genetically engineered Borrelia proteins are used.
The advantage of wholecell lysate Western blots resides in the fact that a greater number of immunoreactive proteins can be detected. The main disadvantage is the difficulty of distinguishing specific from crossreactive bands, and examination by an expert is therefore required.
In recombinant Western blots, specific proteins that can be assigned unambiguously to blot bands are selected. Another advantage is that homologous proteins of different species can be combined [103105]. Recombinant blots have long been considered to have poorer sensitivity than wholecell lysate blots (provided that the latter are performed under standardised conditions with a strain that expresses the diagnostically relevant immunodominant proteins). However, the sensitivity of the recombinant Western blot has now been improved significantly by the addition of recombinant VlsE, a protein that is primarily expressed only in vivo [106].
The decisive criterion qualifying the Western blot as a suitable confirmatory assay is thus reliable identification of the immunoreactive bands. Numerous tests, using antigens of various strains or genospecies of B. burgdorferi sl, are available commercially.
In principle, only tests allowing unequivocal distinction of diagnostically relevant bands should be used. A further prerequisite is reliable specification of these bands by the manufacturer, i.e., based on their identification with monoclonal antibodies (Fig. 6).
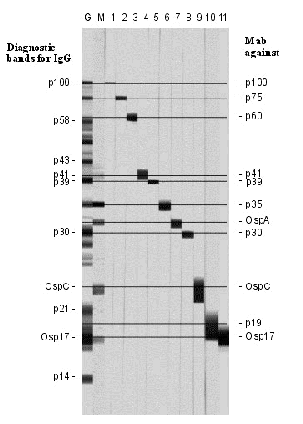
Fig. 6. Standardisation of immunoblots with reference sera.
- G, IgG;
- M, IgM;
- 111, monoclonal antibodies.
- Molecular detection
- Risk assessments: Table 13. Criteria for the diagnosis of Lyme disease based on the EUCALB clinical case definitions for Lyme borreliosis [113,182]
References
92. Wilske B, Schriefer ME. Borrelia. In: Murray PR, Baron EJ, Jorgensen JH, Pfaller MA, Yolken RH, eds. Manual of clinical microbiology. Washington, DC: ASM Press, 2003; 937954.103. Wilske B, Habermann C, Fingerle V et al. An improved recombinant IgG immunoblot for serodiagnosis of Lyme borreliosis. Med Microbiol Immunol 1999; 188: 139144.
104. Roessler D, Hauser U, Wilske B. Heterogeneity of BmpA (P39) among European isolates of Borrelia burgdorferi sensu lato and influence of interspecies variability on serodiagnosis. J Clin Microbiol 1997; 35: 27522758.
105. Wilske B, Fingerle V, PreacMursic V et al. Immunoblot using recombinant antigens derived from different gen-ospecies of Borrelia burgdorferi sensu lato. Med Microbiol Immunol 1994; 183: 4359.
106. Schulte-Spechtel U, Lehnert G, Liegl G et al. Significant improvement of the recombinant Borrelia-specific immu-noglobulin G immunoblot test by addition of VlsE and a DbpA homologue derived from Borrelia garinii for diag-nosis of early neuroborreliosis. J Clin Microbiol 2003; 41: 12991303.
113. Stanek G, OąConnell S, Cimmino M et al. European Union Concerted Action on Risk Assessment in Lyme Borrelio-sis: clinical case definitions for Lyme borreliosis. Wien Klin Wochenschr 1996; 108: 741747.
182. Wilske B, Zo¨ ller L, Brade V et al. MIQ 12 Lyme-Borreliose. In: Mauch H, Lu¨ tticken R, eds. Qualita¨tsstandards in der mikrobiologisch infektiologischen diagnostik.Mu¨ nchen : Urban and Fischer, 2000..
version: 31. March 2004
URL
Home
Joachim Gruber