- [total serum PSA concentration (tPSA)]
[ Shariat SF, Scardino PT, Lilja H, 2008:
- "There is no tPSA threshold at age 62-91 below which prostate cancer (PCa) can be ruled out with high specificity (TN)."
- "No single tPSA cut-off (threshold) separates men with "significant" (high grade,
high volume) cancer from those with low-grade, possibly insignificant cancer."
- "Similar to PCa presence, high-grade cancer can be found in men with low tPSA levels."]
[ Receiver Operating Characteristics (ROC) and verification bias
for test on prostate cancer using the total prostate specific antigen (tPSA) concentration as test parameter. Reference standard: biopsy.
more PSA-ROCs, including proteom analysis by DiaPat)]
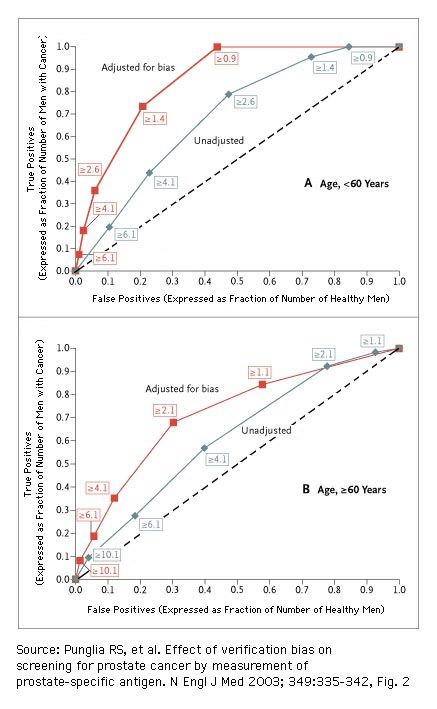
click on figure to get an explanation of this set of ROCs
The numbers in boxes are the PSA threshold values ("test threshold") in ng/mL
The sensitivity (= true positives, expressed as fraction of men with cancer) and 1 - specificity (= false positives, expressed as fraction of healthy men) of a screening test are biased when disease status is not verified in all subjects and when the likelihood of confirmation depends on the test result itself (details). Adjustment for verification bias significantly increased the area under the ROC curve (i.e., the overall diagnostic performance) of the PSA test, as compared with an unadjusted analysis (Punglia et al., 2003)
- AUC = 0.86 after adjustment vs. 0.69 without adjustment, P<0.001, for men less than 60 years of age;
- AUC = 0.72 after adjustment vs. 0.62 without adjustment, P=0.008, for men 60 years of age or older.
[By means of clarification of ROC]
-
A perfect test would have a sensitivity of 1 (in Wikipedia) and a specificity of 1, represented by the point at the upper left-hand corner.
- The curve for a test with no discriminatory power would appear as a diagonal line from the lower left to the upper right corner (the dotted line in the two figures above).
|
|
|
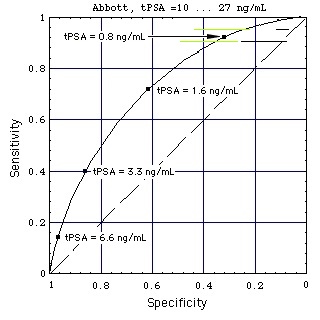
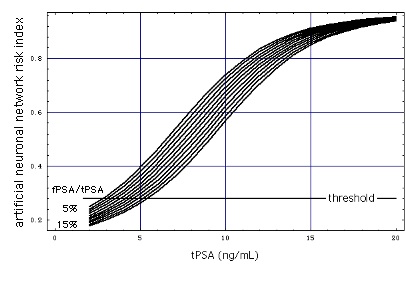
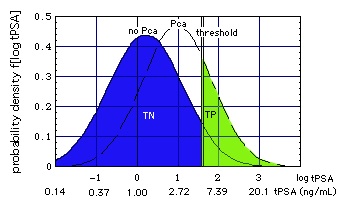
Figure "Lognormal b": Receiver Operating Characteristic (ROC) for lognormal distributions f of tPSA (figure on top of page). tPSA values marked on the ROC are the positions of the thresholds (Punglia et al, 2006, in cache). (Construction of ROC from the distributions)
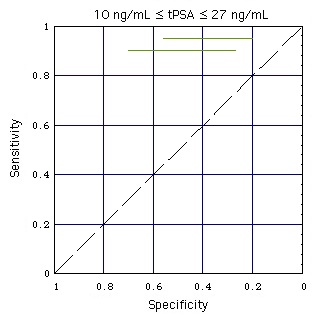
Still Image
- Abbott test kit alone:
In the still image the horizontal black lines show the performance of the Abbott test kit when used for a population of men with high tPSA (10 ... 27 ng/mL).
When a test performance line touches the broken diagonal line, the lognormal tPSA distributions of men with and without Pca coincide (like the tPSA distributions k (with Pca) and g (without Pca) for "increased sex" do in the gray part of this figure).
Result: At high concentrations tPSA looses its diagnostic value.
- Abbott test kit embedded in ANN:
The test performance for a patient population with high tPSA (10 ... 27 ng/mL) is marked with horizontal colored lines. ANN-Data as given in Figure "ANN".
Result: Embedding the tPSA test in the ANN improves the test performance, as the distance of the colored lines from the diagonal is larger than that of the corresponding black lines.
Film: Sensitivity vs. 1 - Specificity
Horizontal lines at sensitivities 0.9 and 0.95 mark the specificity/sensitivity ranges ("performance") of 5 commercial test kits (Stephan et al., 2008, in cache). Reference standard: Biopsy.
- black lines: test kit only (tPSA used as single test parameter, i.e. not in conjunction with other test parameters),
- colored lines: test kit embedded in an artificial neuronal network (ANN).
It is remarkable how much the tPSA test performance decreases above tPSA = 10 ng/mL (black lines are close to the diagonal, at which the test performance is zero).
A reason could be a marked lowering (tentative model) of the blood-prostate-barrier at tPSA > 10 ng/mL. Pca can lower this barrier, but to a similar extent also other effects, such as increased sexual activity, mechanical stress of any kind or an infection can do so.
|
|
- [serum PSA complexes and ratios: test improvements as visualized in ROCs:
- ratios (e.g. free/total PSA concentration ("free-to-total PSA ratio"or "percent free PSA"), PSA density (PSAD, ng/((mL serum) (mL prostate volume)), in cache)),
- complexes (in cache),
- PSA velocity (change of tPSA per year, PSAV, ng/(mL yr))
- see also "Screening for prostate cancer", Shariat SF, Scardino PT, Lilja H, 2008],
- [serum tPSA concentration plus clinical variables: nomograms and artificial neuronal network
- nomograms
- predicting prostate cancer at biopsy (in cache), in Shariat SF, Scardino PT, Lilja H, 2008,
- nomogram to predict indolent cancer, in Kattan et al., 2003 (referred to by Weißbach L and Altwein J, in cache)]
- analysis of the literature, Shariat et al., 2008,
- comparisons of ROCs, Figs. 3 & 4 of Shukla-Dave A et al., 2007,
- Prostate Cancer Nomograms, Memorial Sloan-Kettering Cancer Center Prediction Tools,
|
Figure "Biopsy-detectable":
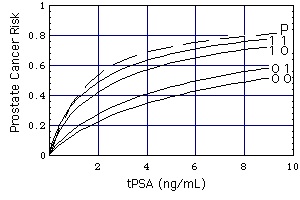
Risk of biopsy-detectable prostate cancer. (Source: University of Texas Health Science Center at San Antonio)
Curves (DRE FAMHIST =) 0 0, 0 1, 1 0, 1 1
Definition of "Prostate Cancer Risk": Take a random sample of 100 men with the characteristics (tPSA, DRE, FAMHIST, PRIORBIOP). "Prostate Cancer Risk" = number of men having Pca in this sample divided by 100. "Prostate Cancer Risk" is calculated from the following nomogram:
Prostate Cancer Risk = 1 / [1 + exp(-PCA)]
with
PCA = -1.7968 + 0.8488 * log(tPSA) - 0.4483 * PRIORBIOP + 0.9054 * DRE + 0.2693 * FAMHIST
Symbols
- PRIORBIOP
- = 0: Past Negative Biopsy: One or more prior biopsies, all negative, no prior diagnosis of prostate cancer.
- = 1: Past Positive Biopsy: Ever a prior positive biopsy or other diagnosis of prostate cancer.
- DRE, FAMHIST
The numbers on the curves specify DRE (left number) and FAMHIST (right number).
- DRE
- =0: normal digital rectal exam,
- =1: abnormal digital rectal exam.
- FAMHIST
- = 0: no family history of prostate cancer,
- = 1: family history of prostate cancer.
Note: This nomogram based calculator is only applicable for persons without a previous diagnosis of prostate cancer.
Curve P is derived from lognormal distributions f of tPSA
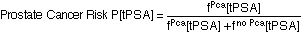
|
- [tPSA variability:
One of the most challenging characteristics of prostate cancer is its variability in biologic aggressiveness, Core needle biopsy specimens are not accurate in the prediction of Gleason score. Studies found that biopsy enabled the correct prediction of radical prostatectomy Gleason grade in
Therefore, the need to improve both tumor detection and assessment of tumor aggressiveness is compelling.
[Fine needle aspiration biopsy DNA ploidy status predicts grade shifting in prostate cancer (Ross et al. 1999).
|
Comparison of the detection of high-grade tumors
by
core needle biopsy histopathological (Gleason) grading and
fine needle aspiration biopsy DNA ploidy analysis
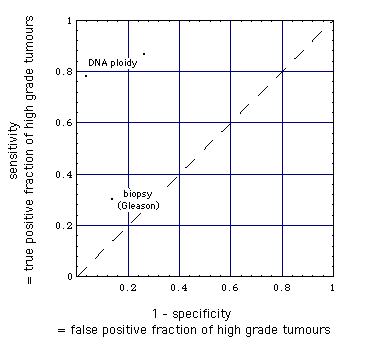
|
The combined use of MR imaging and MR spectroscopy
- improves detection of tumors within the peripheral zone (Males et al. 2000, Scheidler et al, 1999, Obek et al. 1999).
- It has also been shown to increase the specificity in the localization of prostate cancer in the peripheral zone, although with moderate interreader agreement (Scheidler et al, 1999).
- Preliminary reports of MR spectroscopy assessment of tumor aggressiveness have shown promise.
- The ratio of choline and creatine to citrate in the lesion shows correlation with the Gleason grade, with the elevation of choline and reduction of citrate indicative of increased cancer aggressiveness (Zakian et al. 2005).
- It has also been shown that metabolic and volumetric data obtained with MR spectroscopy correlate with the Gleason grade at pathologic examination (Zakian et al. 2005). It has been recommended that the maximum ratio of choline and creatine to citrate combined with tumor volume at MR spectroscopy be used as an index to help predict tumor aggressiveness (Zakian et al. 2003).
MRI versus biopsy: Sensitivities and specificities.
|
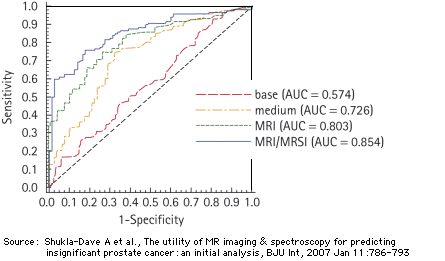
Comparison of the MRI- and the MRI/MRSI models with two models that use a combination of the following test parameters (reference standard for all models: whole mount prostate step section pathology)
- the basis model, presented in the form of a clinical nomogram (mathematical form). It considers
- the total PSA concentration,
- the clinical cancer stadium (cT = T1 ... T3),
- the primary and secondary biopsy Gleason grade,
- the medium model that considers
- the total PSA concentration,
- the clinical cancer stadium (cT = T1 ... T3),
- the primary and secondary biopsy Gleason grade,
- the percentage of the biopsy specimen that turn out cancer positive,
- the prostate volume at the time of the MRI/MRSI.
The MRI and the MRI/MRSI models take into account 6 test parameters:
- the total PSA concentration,
- the clinical cancer stadium (cT = T1 ... T3),
- the primary and secondary biopsy Gleason grade,
- the percentage of the biopsy specimen that turn out cancer positive,
- the prostate volume at the time of the MRI/MRSI,
- an MRI or MRI/MRSI score.
Patient characteristics:
220 patients (cT stage T1c or T2a, prostate-specific antigen level <20 ng/mL, biopsy Gleason score 6, with 41% of the patients having insignificant cancer. Insignificant cancer was defined from surgical pathology as organ-confined cancer of < 0.5 cm3 with no poorly differentiated elements.
Source: Shukla-Dave A, Hricak H, Kattan MW, Pucar D, Kuroiwa K, Chen HN, Spector J, Koutcher JA, Zakian KL, Scardino PT, The utility of magnetic resonance imaging and spectroscopy for predicting insignificant prostate cancer: an initial analysis., BJU Int. 99(4):786-793 (April 2007)
For comparison: AUC for pure MRI and MRSI with biopsy as reference standard: 0.70 and 0.63, respectively (Costouros et al, 2007)
|
Because Gleason grade is an important predictor of patient outcome, this finding provides a rationale for adding MR imaging and/or MR spectroscopy to the pretreatment evaluation of patients with prostate cancer.
[Further Literature, mostly on MR(S)I]
OW Brawley, DP Ankerst, IM Thompson, Screening for Prostate Cancer, CA A Cancer Journal for Clinicians, 2009 (Abstract, local cache of full article)
D Beyersdorff, M Taupitz, B Winkelmann, T Fischer, S Lenk, SA Loening, B Hamm, 2002 "Magnetic resonance (MR), Patients with a History of Elevated Prostate-Specific Antigen Levels and Negative Transrectal US�0�5guided Quadrant or Sextant Biopsy Results: Value of MR Imaging" (in cache), Radiology. 2002 Sep;224(3):701-6.
D Beyersdorff, A Winkel, B Hamm, S Lenk, SA Loening, 2005 "MR Imaging-guided Prostate Biopsy with a Closed MR Unit at 1.5 T: Initial Results" (in cache), Radiology. 2005 Feb;234(2):576-81. Epub 2004 Dec 22.
PR Carroll, FV Coakley, and J Kurhanewicz, 2006 "Magnetic Resonance Imaging and Spectroscopy of Prostate Cancer" (in cache), Radiology. 2006 Nov;241(2):546-53.
FG Claus, H Hricak, RR Hattery, 2004 "Pretreatment Evaluation of Prostate Cancer: Role of MR Imaging and 1H MR Spectroscopy" (in cache), Radiographics. 2004 Oct;24 Suppl 1:S167-80.
FV Coakley, I Chen, A Qayyum, AC Westphalen, PR Carroll, H Hricak, M Chen and J Kurhanewicz, 2006
"Validity of prostate-specific antigen as a tumour marker in men with prostate cancer managed by watchful-waiting: correlation with findings at serial endorectal magnetic resonance imaging and spectroscopic imaging (in cache), BJU Int. 2007 Jan;99(1):41-5.
NG Costouros, FV Coakley, AC Westphalen, A Qayyum, BM Yeh, BN Joe, J Kurhanewicz, "Diagnosis of prostate cancer in patients with an elevated prostate-specific antigen level: role of endorectal MRI and MR spectroscopic imaging, AJR Am J Roentgenol. 2007 Mar;188(3):812-6.
H Ito, K Kamoi, K Yokoyama, K Yamada, T Nishimura, "Visualization of prostate cancer using dynamic contrast-enhanced MRI: comparison with transrectal power Doppler ultrasound" (in cache), Br J Radiol. 2003 Sep;76(909):617-24.
MW Kattan, JA Eastham, TM Wheeler et al., "Counseling men with prostate cancer: a nomogram for predicting the presence of small, moderately differentiated, confined tumors. J Urol 2003Nov;170(5):1792-7 (referred to by Weißbach L and Altwein J, in cache)
D Pucar, A Shukla-Dave, H Hricak, CS Moskowitz, K Kuroiwa, S Olgac, LE Ebora, PT Scardino, JA Koutcher, 2005 "Prostate Cancer: Correlation of MR Imaging and MR Spectroscopy with Pathologic Findings after Radiation Therapy: Initial Experience (in cache), Radiology. 2005 Aug;236(2):545-53. Epub 2005 Jun 21
A Qayyum, FV Coakley, V Lu1, JD Olpin, L Wu, BM Yeh, PR Carroll and J Kurhanewicz, 2005 "Organ-Confined Prostate Cancer: Effect of Prior Transrectal Biopsy on endorectal MRI and MR Spectroscopic Imaging" (in cache), AJR Am J Roentgenol. 2004 Oct;183(4):1079-83.
SF Shariat, PI Karakiewicz, N Suardi, MW Kattan, "Comparison of nomograms with other methodes for predicting outcomes in prostate cancer: a critical analysis of the literature", Clin Cancer Res 2008;14(14):4404-4407, July 15, 2008 (in cache)
SF Shariat, PT Scardino, H Lilja, "Screening for prostate cancer: an update", Can J Urol 2008;15(6):4363-4374, December (in cache)
A Shukla-Dave, H Hricak, SC Eberhardt, S Olgac, M Muruganandham, PT Scardino, VE Reuter, JA Koutcher, 2004 "Chronic Prostatitis: MR Imaging and 1H MR Spectroscopic Imaging Findings - Initial Observations" (in cache), Radiology. 2004 Jun;231(3):717-24.
A Shukla-Dave, H Hricak, MW Kattan, D Pucar, K Kuroiwa, HN Chen, J Spector, JA Koutcher, KL Zakian, PT Scardino. 2007 "The utility of magnetic resonance imaging and spectroscopy for predicting insignificant prostate cancer: an initial analysis." (in cache), BJU Int. 2007 Apr;99(4):786-93. Epub 2007 Jan 12.
KL Zakian, S Eberhardt, H Hricak, A Shukla-Dave, S Kleinman, M Muruganandham, K Sircar, MW Kattan, VE Reuter, PT Scardino and JA Koutcher, 2003 "Transition Zone Prostate Cancer: Metabolic Characteristics at 1H MR Spectroscopic Imaging - Initial Results" (in cache), Radiology. 2003 Oct;229(1):241-7. Epub 2003 Aug 14.
KL Zakian, K Sircar, H Hricak, HN Chen, A Shukla-Dave, S Eberhardt, M Muruganandham, L Ebora, MW Kattan, VE Reuter, PT Scardino and JA Koutcher, 2005 "Correlation of Proton MR Spectroscopic Imaging with Gleason Score Based on Step-Section Pathologic Analysis after Radical Prostatectomy" (in cache), Radiology. 2005 Mar;234(3):804-14.
Appendix
T Primary Tumor
TX: Primary tumor cannot be assessed
T0: No evidence of primary tumor
T1: Clinically inapparent tumor not palpable or visible by imaging
T1a: Tumor incidental histologic finding in > 5% of tissue resected via TURP
T1b: Tumor incidental histologic finding > 5% of tissue resected via TURP
T1c: Tumor identified by needle biopsy (e.g., because of elevated PSA)
T2: Tumor palpable but confined within the prostate
T2a: Tumor involves half of a lobe or less
T2b: Tumor involves more than half a lobe, but not both lobes
T2c: Tumor involves both lobes
T3: Tumor extends through the prostatic capsule
T3a: Unilateral extracapsular extension
T3b: Bilateral extracapsular extension
T3c: Tumor invades the seminal vesicle(s)
T4: Tumor is fixed or invades adjacent structures other than the seminal vesicles
T4a: Tumor invades any of bladder neck, external sphincter or rectum
T4b: Tumor invades levator muscles and/or is fixed to the pelvic wall
N Regional Lymph Nodes
NX: Regional lymph nodes cannot be assessed
N0: No regional lymph nodes metastasis
N1: Metastasis in a single lymph node, 2 cm or less in greatest dimension
N2: Metastasis in a single lymph node, more than 2 cm but not more than 5cm in greatest dimension; or multiple lymph node metastases, none more
than 5 cm in greatest dimension
N3: Metastasis in a lymph node more than 5 cm in greatest dimension
M Distant Metastases
MX: Presence of distant metastasis cannot be assessed
M0: No distant metastasis
M1: Distant metastasis
M1a: Nonregional lymph node(s)
M1b: Bone(s)
M1c: Other site(s)
Although randomized controlled studies are lacking, current data of active surveillance permit the conclusion that doctors have an obligation to alert patients to this option [of active surveillance]. Current guidelines on the treatment of PCa [prostate cancer] also confirm that the treatment of localized prostate cancer has undergone a rethink. However, this is barely reflected in clinical practice, at least not in Germany. In spite of the shift in tumor stage to earlier diagnosis as described earlier, the number of radical prostatectomies is rising steadily, particularly in the group of patients for whom active surveillance as laid out in the latest guidelines would be a suitable option. According to 2006 DRG statistics, almost 69% of men younger than 70 undergo RP. The numbers of operations have risen in recent years. The Brandenburg cancer registry shows that the following therapies were applied in men younger than 70 with tumor categories pT1 to pT3, for 2003 - 2005:
- Radical prostatectomy in 70% of patients
- Exclusively radiotherapy in 15% of patients
- Defensive strategies such as hormone therapy, WW [Watchful Waiting], or active surveillance were used in only 15% of patients.
Almost two thirds of PCa found were at stage T1 or T2 and would have been suitable for active surveillance [Bandemer-Greulich U, Wulff V, Marquaß S, Kindt B, "7.16 Prostatakarzinome (C61)" in Qualitätsbericht Onkologie 2009, Tumorzentrum Land Brandenburg e.V. und Qualitätskonferenz Onkologie (QKO), 2009 (in cache)].
There are no more precise data from Germany that show how many patients have a low risk PCa and would therefore be suitable to undergo active surveillance. Currently the best data source on PCa is the longitudinal data collection of CaPSURE. Of more than 10 000 patients included between 1989 and 2003,
- 29.7% had low risk tumors, for which NICE recommends active surveillance as the first therapeutic option.
- In 1989/90, 31% were in this risk group, but the proportion rose to 47% in 2001/2002.
- Over the same time period, the proportion of high risk tumors fell from 41% to 15%.
These data give rise to the assumption that in Germany, the 69% of prostatectomies in men younger than 70 include many cases with an excellent prognosis.
PCa treatment should aim to provide each patient with the treatment that is appropriate for his personal needs, individual medical history, and tumor biology. Radical, risk prone interventions should be considered if they are unavoidable and the patient's survival gain justifies the risks associated with the intervention.
In the range of options, active surveillance is the strategy that enables risk assessment and making considered, unhurried therapeutic decisions: a retrospective controlled study with 188 participants (level of evidence 2b) showed that radical prostatectomy delayed by 26 months does not impair the chances of curative treatment in small and well differentiated tumors if they
- are category T1c,
- have a PSA density <0.15 ng/mL/cm,sup>3,
- have a Gleason score <7, and
- are detectable in no more than two positive biopsy cores with less than 50% of tumor mass in each core.
Knowledge of the tumor biology and giving appropriate consideration to all available treatment strategies can enable primary care physicians to become important decision-makers jointly with their patients. Some food for thought: an editorial, suitably entitled "Prostate Cancer: are we over-diagnosing - or under-thinking?", concludes with the following advice: "Think more!".
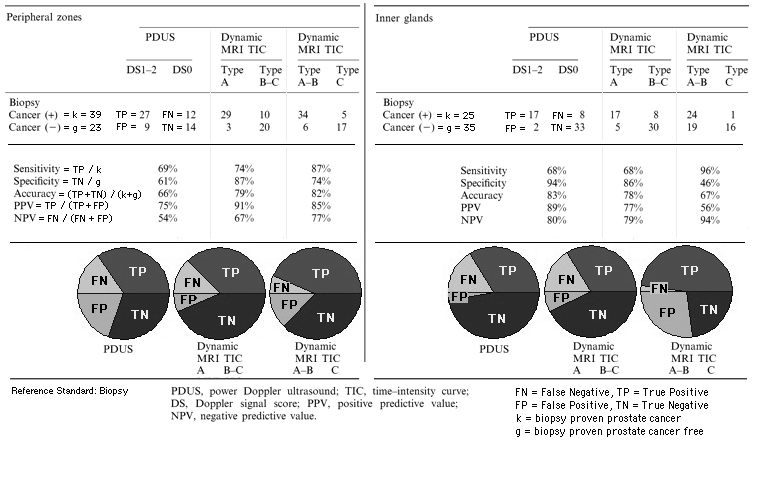
Both
- transrectal power Doppler ultrasound (PDUS, ultrasound machine: SSD-5500, Aloka, Tokyo, Japan, endorectal biplanar probe: convex 5 MHz and linear 7.5 MHz) and
- dynamic contrast-enhanced Magnetic Resonance Imaging (DCE-MRI, 1.5 T superconducting MR system: Gyroscan Intera, Philips Medical Systems, Best, The Netherlands)
can be used to demonstrate hypervascularity in many prostate cancers. DCE-MRI was significantly more sensitive than PDUS for visualizing of prostate cancers without loss of specificity in the peripheral zone.
Version: 17. August 2012
Address of this page
Home
Joachim Gruber


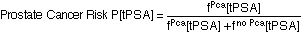
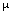 ,
, 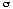 ]
]