Home -- Symptoms -- Cycles -- Cefepime Model Calculations

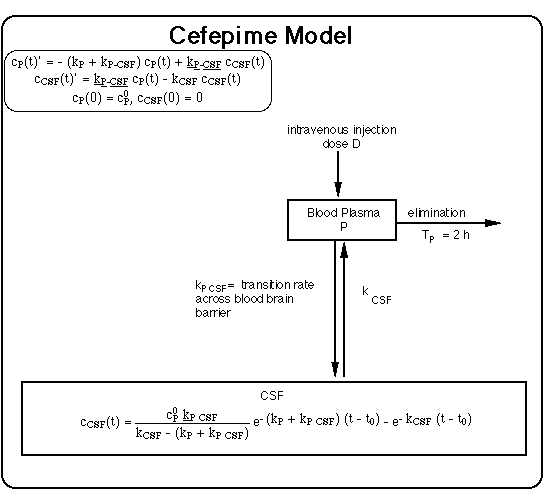
Fig. 1: Model for cefepime pharmacokinetics used to calculate cefepime concentration in CSF. Corresponding linear set of differential equations is shown in top of Figure. Equation in CSF compartment is analytical solution to this set. Notation: concentrations, rate constants k, t0.
Fig. 2: Fit of pharmacokinetic model (curves) to reproduce total cefepime concentrations in serum and CSF (points and circles). Elimination half life of CSF compartment is Tcsf = ln2/kcsf = 1 hour. Time dependent levels in CSF were calculated for two lag times: tau = 0 (light curve) and tau = 1 h (heavy curve). Points: data published in Bristol Myers Squibb Cefepime Presciption Information. Circles: two samples taken after 6 infusions of 2 g cefepime each, distance between infusions: 8 h, samples were drawn 5 h after last infusion. Upper circle: level in serum (19 mg/L), lower circle: level in CSF (0.75 mh/L). Samples courtesy of Dr. Patricia Coyle, Department of Neurology, State University of New York, Stony Brook, N.Y. 11794, USA. HPLC determination of levels in samples courtesy of Herman Mattie, Department of Infectious Diseases, University Hospital Leiden, NL - 2300 RC Leiden, The Netherlands.
The model paramters used in the pharmacokinetic calculations are
where data from Bristol Myers Squibb have been printed in plain font, and the data from model fit shown in Fig. 2 in underlined font. Bold face values denote the conservative estimate.
Note that the tau = 1 case (heavy curve) results in a higher post peak concentration in the CSF as the tau = 0 case (light curve). The latter fit will thus be called a "conservative estimate".
Fig. 3 shows a fit of the data with the same values for TP = 2.0 h and cP(0) = 100 mg/L
Fig. 3: Fit of pharmacokinetic model (curves) to reproduce total cefepime concentrations in serum and CSF (points and circles). Elimination half life of CSF compartment is Tcsf = ln2/kcsf = 2 hours, otherwise data and notation as in Fig. 2.
Note that this fit of data results in a higher cefepime concentration in the CSF than the fit for TCSF = 1.0 h. The same is true for the tau = 1 h fit here as with the previous set of parameters (Figs. 2 and 3). Thus, the fit with
Fig. 4: Systematics of MIC's for a range of antibiotics (horizontal axis) and gram negative bacilli. Vertical axis: logarithm of MIC, MIC measured in mg/L.Classification ("penicillin susceptible", etc) from Spangler 1997).
The beta-lactam antibiotics used in this systematics consist of one carbapenem (imipenem) and 4 cephalosporins. For the listed bacteria, the MIC of cefepime is generally more than a factor of 2 smaller than the MIC of the rest of the cephalosporins, an exception being Moraxella catarrhalis. The data from Bristol-Myers Squibb and Preac Mursic et al. are basically consistent.
In the following evaluations the MIC range provided by Bristol-Myers Squibb will be used.
 |
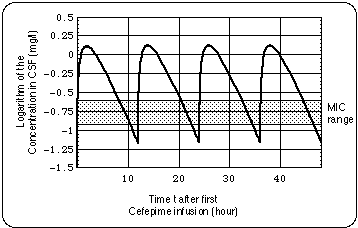 |
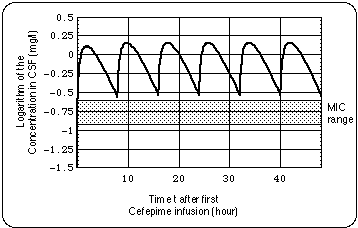
When 2 grams of cefepime are infused only every 12 hours, the concentration in the CSF falls below the range 0.26 ... 0.12 mg/L in which the Minimum Inhibitory Concentration for Bb is expected to lie. When the interval between infusions is 8 hours, the cefepime level stays well above that range.
ln2 = natural logarihm of 2 = 0.69.
Kessler RE, Fung-Tomc J, Susceptibility of bacterial isolates to beta-lactam antibiotics from U.S. clinical trials over a 5-year period, Am J Med 1996;100(suppl 6A):13S-19S.
Samples from patient whose data were analyzed in J. Gruber, "Lyme Disease: Statistical Evaluation of a Symptom Log and an Empirical Theory of Flare Cycles".
Spangler SK, Jacobs MR, Apfelbaum PC,MIC and Time-Kill Studies of Antipneumococcal Activity of 118819X (Sanfetrinem) Compared with Those of Other Agents, Antimicrob Agents Chemother 1997; 41:141-155
Thornsberry et al. In vitro activity of cefepime and other antimicrobials: survey of European isolates, J. Antimicrobial Chemother. 1993, 32, Suppl. B, 55-62.
Wagner D private communication, Pharmaceutical Info Div., 777 Scudders Mill Road, Plainsboro, NJ 08536, Tel.: +1 (609) 897 - 2776, Fax: +1 (609) 897 - 6042.
version: December 17, 1998
Send comments to Joachim Gruber